Title: Heterochiral coupling to bilateral β-turn structured azapeptides bearing two remote chiral centers
Authors: Xiaosheng Yan, Jinlian Cao, Huan Luo, Zhao Li, Zexing Cao, Yirong Mo & Yun-Bao Jiang*
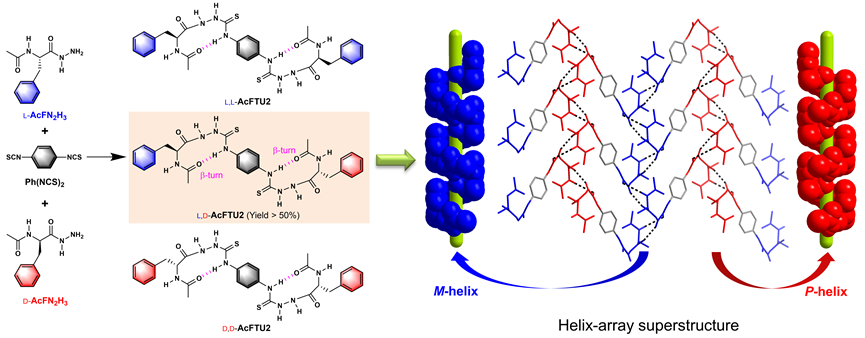
Abstract: Enantioselective synthesis governed by chiral catalysts has been extensively developed, but that without any chiral auxiliaries or chiral catalysts is rare, particularly when remote stereogenic centers are involved. Here we report an enantioselectivity of heterochiral coupling in the one-pot reaction of racemic hydrazides with achiral 1,4-bis(isothiocyanine)benzene, yielding preferentially the heterochiral bilateral azapeptides over the homochiral ones. Despite bearing two hydrogen-bonded β-turn structures that allow intramolecular chiral transfer, the bilateral azapeptide products have two chiral centers separated by 14 atoms or 15 bonds, which prevent the direct intramolecular asymmetric communication between the two chiral centers. Interestingly, the heterochiral azapeptides feature intermolecular hydrogen bonding stacking between homochiral β-turns to form a superstructure of alternative M- and Phelices in the crystals. In contrast, the homochiral azapeptide counterparts adopt a β-sheet-like structure, which is less favorable compared to the helicallike superstructure from heterochiral azapeptides, accounting for the favored heterochiral coupling of the one-pot reaction. This work demonstrates enantioselective synthesis involving distant chiral centers through the formation of biomimetic superstructures, opening up new possibilities for the regulation of enantioselectivity.
Full-Link: https://www.nature.com/articles/s41467-024-53744-x